- Laboratory >
- Laboratory medicine >
- Rapid respiratory disease test
Rapid respiratory disease tests
Result display time: 20 min
Sample volume: 0.03 ml
Specificity: 100 %
... testing results than any other laboratory-based methods with higher accuracy than conventional rapid testing. With less than a minute of hands-on time, the test allows the detection of SARS-CoV-2 in 20 minutes from sample ...
Credo Diagnostics Biomedical Pte Ltd
... results cannot be guaranteed if there are any deviations from the instructions for use. The test is for Professional Use Only. PACKING SPECIFICATION 20 Tests/ Kit INTENDED USE This kit is used for in vitro ...

... Multiplex nucleic acid detection kit for Influenza A virus, Influenza B virus and 2019-nCoV Compositions: Buffer, Enzyme Mix.Positive & Negative control Size: 48/96 T/Kit Storage: -20±5°C Shelflife: 12 months ...
Genrui Biotech Inc.

Result display time: 15 min
... Influenza B Rapid Test is used for qualitative detection of COVID-19, Influenza Type A and B nucleoproteins antigens in human Nasal, Nasopharyngeal and Oropharyngeal swabs samples. b. PACKAGE 1 tests ...
Beijing O&D Biotech Co., Ltd.
... with low immune function. It has been proven that 80% of acute upper respiratory diseases and most lower respiratory diseases are caused by pathogens outside of bacteria, with respiratory ...
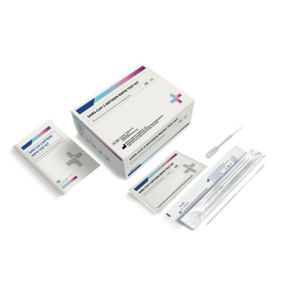
Result display time: 15 min
Specificity: 99.6, 98.9 %
Sensitivity: 97.7, 96.8 %
... KIT COMPONENTS 25 Test Cassettes 25 Pipettes 25 Swabs 2 Extraction Buffer (4mL) 25 ExtractionVials/Caps 1 Instruction For Use SPECIFICATIONS Storage Temperature: 2-30ºC Shelf life: 12 months ...

Result display time: 3 min
... in identifying individuals with an acute SARS-CoV-2 infection. VYRA™CoV2Flu is a combination viral nucleocapsid antigen tests that detects infection caused by SARS-CoV-2 and Influenza A (Flu A) and Influenza B (Flu B). VYRACoV2Flu ...

Result display time: 20 min - 30 min
Specificity: 100 %
Sensitivity: 90.6 %
... Specimen Volume 3 drops Storage Condition 2~30℃ (36~86℉) Pack Size 25 tests/Box ...
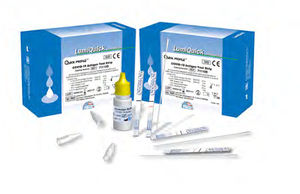
... QuickProfile™ COVID-19 Antigen Test is a rapid in vitro immunochromatographic assay for the qualitative detection of SARS-CoV-2 virus antigen present in human nasopharynx COLLECTION: Proper specimen collection, ...
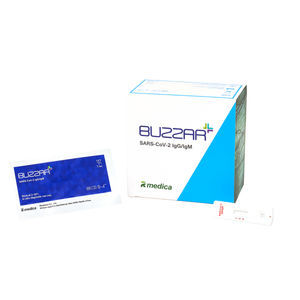
Result display time: 15 min
Specificity: 100 %
Sensitivity: 98 %
... as 1/8 Analytical Specificity ① No cross-reaction against 25 antisera of respiratory disease ② No difference in specificity according to 14 interference materials Test method The sample pad ...

... Rapid qualitative test that detects influenza type A and type B. 25 Tests per box Description Rapid qualitative test that detects influenza type A and type B Includes: Positive ...

Result display time: 10, 15 min
... Fortress Diagnostics have developed a range of tests in their efforts to help respond to the requirement for reliable detection of Covid-19. ...

... Specifications Strep A Test Cassette Format: cassette Specimen: nasal swab Model No:STA-S02D High Accuracy OEM available Strep A Test Cassette PRINCIPLE OF THE TEST Group A Streptococci (S. pyogenes) ...

Result display time: 15 min
Specificity: 100 %
Sensitivity: 87.3 %
... The Innovita COVID-19 IgG/IgM Rapid Test is designed for the qualitative detection of IgG and IgM antibodies directed against SARS-CoV-2 from whole blood, serum and plasma samples. The test provides a ...

Specificity: 100 %
Sensitivity: 95 %
... Test Type - Qualitative Storage Condition - All components of this kit can be stored at 2℃-30℃. Shelf-life - 24 months Target patient - Human ...

Result display time: 15 min
Specificity: 99.3 %
Sensitivity: 96.3 %
... equipment. resuits are clearly visible. Convenient transportation and low price, higher accuracy. Suitable for large-sacle rapid screening. Easy operation Sample collection Open the cover of the saliva collection tube. Screw ...
Jiangsu Konsung Medical Equipment

Result display time: 15 min - 20 min
Sample volume: 0.01, 0.015 ml
Specificity: 98.2 %
... Temperature - 2 – 30 °C Package Includes - Cassette Disposable specimen applicator Buffer Package Insert Tests / Kit - 25 Tests / Kit ...

Result display time: 10 min - 15 min
Sample volume: 0.08 ml
... other viruses infecting respiratory track have similar symptoms, it is critical to properly diagnose influenza viral infection before any treatment. Flu A&B Plus differentiates influenza A and B diagnosis with boldly colored test ...

... weakened immune systems. HMPV is associated with colds, bronchiolitis, pneumonia, and upper respiratory tract infections. The virus spreads through respiratory droplets and is most common during winter and spring seasons. ...

Result display time: 30 min
... Unsure if your respiratory symptoms, such as fever, sneezing, cough, or fatigue, are from COVID? With our at-home rapid test, quickly find out and get the right care for you and your family. Simple ...
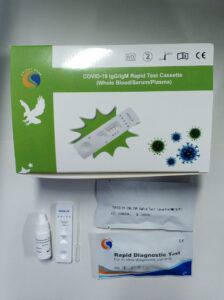
... COVID-19 IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma) is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG/IgM to 2019 Novel Coronavirus ...

Result display time: 10 min - 15 min
... COVID-19 Antigen Self- Test is a single-use, in vitro (outside the body) rapid immunochromatographic test that uses a human nasal swab specimen for the qualitative detection of nucleocapsid protein antigen ...

Result display time: 15 min
Sample volume: 0.075 ml - 0.1 ml
... EASY TESTING A Simple Nasal Swab CONVENIENT No equipment required Specification: Storage Condition 2℃- 30℃ Shelf Life 18 months Packing Size 1pc/kit, 2pcs/kit, 5pcs/kit, 25pcs/kit ...
Bioscience (Tianjin) Diagnostic Technology Co.,Ltd

Result display time: 15 min
Sample volume: 0.1 ml
Specificity: 99 %
... The test is a lateral flow immunoassay designed to detect SARS-CoV-2 viral antigens in a swab sample. This test is intended for use in individuals to suspect a COVID-19 infection. The REDCELL COVID-19 Self Test ...

Result display time: 10 min
Specificity: 99.2 %
Sensitivity: 91.6 %
... swab (10 ea) • Disposable dropper (10 ea) PACKAGES • 10 Tests/Kit • 12 Kits/Box (120 Tests/Box) or 40 Kits/Box (400 Tests/Box) TEST PROCEDURE 1. All reagents and samples must ...
the best suppliers
Subscribe to our newsletter
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group