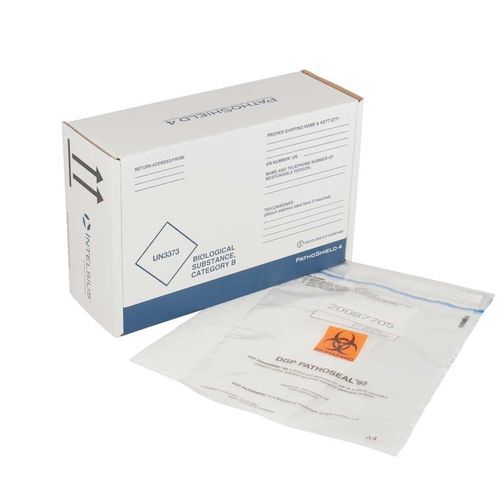
The PathoSeal 95 range is designed and tested to withstand a pressure difference of 95 kPa to meet UN3373 Biological Substance, Category B regulations.
The PathoSeal Liquitite range is designed and tested to meet Category B transport regulations when used in conjunction with 95 kPa primaries.
This range of flexible secondary packaging is ideal for the transportation of a variety of primary containers such as blood vials, swabs (including COVID-19 test swabs), blood collection bags and medical devices.
All PathoSeal products feature a simple closure system enabling ease of packing, plus the unique tamper evident seal ensures the integrity of samples. In addition the bags are sequentially numbered, providing a chain of custody and additional security. An external document pouch is also present for all sizes with the exception of PathoSeal 95 A6.