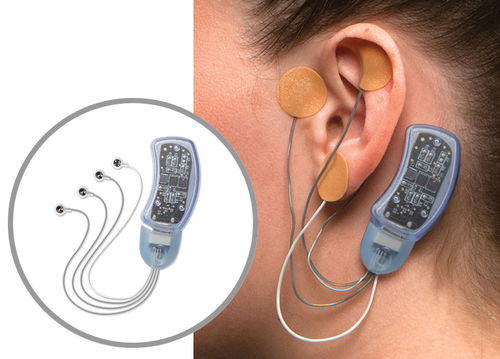
Transcutaneous neurostimulator Bridge™for auricular vagus nerve stimulation

Add to favorites
Compare this product
fo_shop_gate_exact_title
Characteristics
- Type of neurostimulator
- transcutaneous
- Type of neurostimulation
- for auricular vagus nerve stimulation
Description
The first FDA-cleared medical device for the reduction of opioid withdrawal symptoms,* Bridge is designed to give your patients the hope they need to continually progress through treatment—without the fear of withdrawal.
Proven to Provide Relief
FAST
85% reduction in the severity of a patient’s symptoms in the first hour of placement, with the first signs of relief in as few as 20 minutes
EFFECTIVE
88% of patients successfully transitioned into a medication-assisted treatment (MAT) program while using Bridge
97% reduction in reported withdrawal symptom severity by the fifth day of wear
SAFE
0 reported side effects with minimal risks during device placement
How Does Bridge Work?
Bridge is a small electrical nerve stimulator device that contains a battery-powered chip and wires that are applied around a patient's ear in a short, non-surgical, in-office procedure, providing five days of continuous relief.†
Applied by a qualified healthcare professional in a short, non-surgical, in-office procedure
Fits comfortably behind the ear
Provides five days of continuous relief
Stimulated nerves transmit these impulses to the brain, reducing withdrawal symptoms
Sends gentle electrical impulses through wires to nerves around the ear
VIDEO
Catalogs
No catalogs are available for this product.
See all of Masimo‘s catalogsRelated Searches
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.

