- Laboratory >
- Immunology >
- Antibody test kit
Antibody test kits

Result display time: 10 min - 12 min
PolyStacker™ Plus Detection Kits is specially designed for intraoperative frozen immunohistochemistry detection technology. The kits can be used for the detection for the chromogen of ...



... Intended use This ELISA PathRF kit is intended for the quantification of human IFN-γ in serum and plasma containing rheumatoid factor (RF). For research use only. Not for use in diagnostic procedures. ELISA ...

... Intended use This ELISA PathRF kit is intended for the quantification of human IL-10 in serum and plasma containing rheumatoid factor (RF). For research use only. Not for use in diagnostic procedures. ELISA ...

... Intended use This ELISA PathRF kit is intended for the quantification of human IL-8 (CXCL8) in serum and plasma containing rheumatoid factor (RF). For research use only. Not for use in diagnostic procedures. ELISA ...

Result display time: 60 min
Sample volume: 0.01 ml
The CytoBead® ANCA is an immunofluorescence assay (IFA) for the qualitative and semi-quantitative determination of IgG antibodies in human serum against neutrophil cytoplasmic antigens (ANCA) and the glomerular basal ...
MEDIPAN GmbH

Result display time: 80 min
... vessel walls, and the resulting morphological changes. Anti-neutrophil cytoplasmic antibodies (ANCA) play an essential role in the serological diagnosis of SV. These antibodies are usually determined ...
MEDIPAN GmbH

Result display time: 80 min
... immunofluorescence) on neutrophils. The occurrence of antibodies to mitochondrial antigens (M2) is typical of PBC. AIH show different autoantibodies, especially autoantibodies to; antinuclear antibodies ...
MEDIPAN GmbH
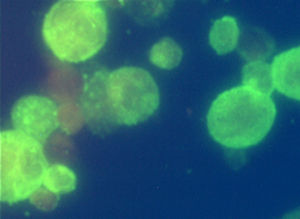
... inflammatory mono- and polyneuropathies). The diagnostic kits can be also utilized for differential diagnosis of neuroinfections, infections of eye and skin and exanthematous diseases. The tests do not ...

Result display time: 20 min
Gmate COVID-19 Neutralizing antibody kit is an in vitro diagnostic medical device that qualitatively detects neutralizing antibodies to COVID-19 in patient's blood by immunochromatography Specification Specimen ...

This kit is used to quantitatively detect the content of rheumatoid factor in human serum in vitro. Background of Clinical Indications Increased: seen in patients with rheumatoid arthritis, systemic lupus erythematosus ...

Specificity: 91 %
Sensitivity: 57 %
Specificity: 100 %
Sensitivity: 97.7 %
Specificity: 98.1 %
Sensitivity: 92.9 %

Result display time: 10 min
... in minutes in order to enable accurate clinical decisions. Improving access : Rapid diagnostic tests facilitate access to healthcare globally. Content: 20 tests Also available for automated reading ...

Result display time: 10 min
... in minutes in order to enable accurate clinical decisions. Improving access : Rapid diagnostic tests facilitate access to healthcare globally. Content: 10 tests

Sample volume: 0.05, 0.03, 0.1 ml
... determination of TSH is the first choice to judge whether the thyroid and hypothalamic-pituitary-thyroid axis function is normal. T3 test can effectively assess thyroid function:(1) diagnosis and early diagnosis of ...
Autobio Diagnostics

The measurement of anti-CCP antibodies has become the method of choice in the accurate diagnosis of Rheumatoid Arthritis. Assays using the second-generation CCP2 peptide are as sensitive as the rheumatoid factor (RF) ...
Axis-Shield PLC

... are very important markers for thyroiditis and either one or both appear in 98% of thyroiditis patients. All our thyroid kits are calibrated against the international standards and provide you with accurate results ...
AESKU.GROUP

Specificity: 97.5 %
Sensitivity: 81.8 %
The Inhalant-Food Allergen-specific IgE Antibodies Test Kit is qualitative, membrane based Immunoblotting for detection of antibodies to allergen-specific IgE in human ...
Result display time: 15 min
Sample volume: 0.02 ml
... line on the test region (T), indicating a positive result. Control line should always appear if the test procedure is performed properly and the test reagents are ...

Result display time: 60, 30, 15 min
Sample volume: 0.01 ml
... quantitative determination of IgG antibodies to cytochrome P450 IID6 in human serum or plasma. Anti-LKM-1 is an enzyme immunoassay for the quantitative determination of IgG antibodies to cytochrome P450 ...
DRG Instruments GmbH

... transfusion accident is potentially DEADLY. The final pre-transfusion test is the last step to detect a failure in the transfusion circuit and, among other things, this test prevents an ABO haemolytic ...

Manufactured by SafePath® exlusively for Centaur Animal Health, the FP-ELISA II Kit is Centaur’s third generation test for antibodies to Equine Infectious Anemia Virus in horse serum. ...
Result display time: 15 min - 20 min
Specificity: 99.1 %
Sensitivity: 98.3 %
The SARS-CoV-2 Neutralization Antibody Rapid Test Kit is a lateral flow chromatographic immunoassay for the qualitative detection of neutralization antibody to severe ...

This diagnostic kits are based on RealTime PCR method. In this kits primers and labeled probes (FAM and JOE) for the detection of alleles and for the detection of internal control are used. Compatible ...
... staining of circulating epithelial cells in peripheral blood. Cells are captured using anti-EpCAM antibodies bound to ferrofluid. Fluorescent staining reagents are then added for identification of ...

Result display time: 60 min
... Detection Colorimetric The Anti-HAV IgG ELISA is an enzyme immunoassay for the qualitative detection of the Hepatitis A virus antibody (Anti-HAV) in human serum or plasma. The Anti-HAV IgG ...
ALPCO

The Biomerica anti-LKM1 (Liver Kidney Microsome Type 1) antibodies kit is a quantitative enzyme immunoassay for the detection of autoantibodies to LKM1 in human serum.

In these uncertain times, laboratories need to have confidence in COVID-19 antibody test results—which is why Luminex has developed a comprehensive SARS-CoV-2 antibody assay. Using a ...

Result display time: 15 min
... Neutralizing Antibody Rapid Test is intended for in vitro qualitative detection of SARS-CoV-2 neutralizing antibodies in human serum, plasma, venous whole blood or fingertip blood. The ...
Getein Biotech

Vasculitis (ANCA-PR3, ANCA - MPO, GM) Regulatory Status (USA):510(k) Cleared Discipline:Autoimmune Vasculitis (bulk pack) Regulatory Status (USA):510(k) Cleared Discipline:Autoimmune
Gold Standard Diagnostics
Your suggestions for improvement:
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group


























Please specify:
Help us improve:
remaining