- Laboratory >
- Laboratory medicine >
- Influenza B test kit
Influenza B test kits
{{product.productLabel}} {{product.model}}
{{#if product.featureValues}}{{product.productPrice.formattedPrice}} {{#if product.productPrice.priceType === "PRICE_RANGE" }} - {{product.productPrice.formattedPriceMax}} {{/if}}
{{#each product.specData:i}}
{{name}}: {{value}}
{{#i!=(product.specData.length-1)}}
{{/end}}
{{/each}}
{{{product.idpText}}}
{{product.productLabel}} {{product.model}}
{{#if product.featureValues}}{{product.productPrice.formattedPrice}} {{#if product.productPrice.priceType === "PRICE_RANGE" }} - {{product.productPrice.formattedPriceMax}} {{/if}}
{{#each product.specData:i}}
{{name}}: {{value}}
{{#i!=(product.specData.length-1)}}
{{/end}}
{{/each}}
{{{product.idpText}}}

Result display time: 75 min
Real-time RT-PCR test for the qualitative detection of HA gene from Influenza A/B virus and N and ORF1a gene from SARS-CoV-2 in nasopharyngeal swab. Details Features: Hot Start ...

Result display time: 15 min - 20 min
This product is used for qualitative detection of 2019-nCoV antigen, Influenza A virus antigen and Influenza B virus antigen in clinical samples. 2. Product Features 1 Sample, 3 ...

... analyses. The list of wet-lab-tested respiratory pathogens includes Influenza A (H3N2, H1N1), Influenza B, Parainfluenza (1-4), Coronavirus 229E, OC43, NL63, C. pneumoniae, Metapneumovirus,B. ...

... Virus Nucleic Acid Multiplex Detection Kit Cat. No : P740H Specification: 25T/kit(non-prefilled) Specimen : Nasopharyngeal or oropharyngeal swabs, sputum, alveolar lavage fluid Target ...
Xian Tianlong Science and Technology

... This kit is used for qualitative detection of respiratory virus nucleic acids in clinical samples. Respiratory virus (RV) is a virus that causes respiratory infections in humans. This kit can ...
Xian Tianlong Science and Technology

... This kit is used for qualitative detection of respiratory virus nucleic acids in clinical samples. Respiratory virus (RV) is a virus that causes respiratory infections in humans. This kit can ...
Xian Tianlong Science and Technology
Result display time: 10 min
... highly contagious respiratory disease but triggered by different virus: SARS-CoV-2 and Flu A&B, respectively. GOLDSITE SARS-CoV-2 & Influenza A/B Antigen Kit (Colloidal ...
Result display time: 15 min
... like to determine what type of infection you have: SARS-CoV-2, influenza A/B, and/or respiratory syncytial virus (RSV).GOLDSITE SARS-CoV-2 & Influenza A/B & RSV Antigen ...
Result display time: 15 min
... from influenza and help prevent the spread of disease in the population. Diagnostic: Influenza A/B Antigen Kit GOLDSITE Influenza A/B ...

Result display time: 3 h
... Aptima SARS-CoV-2/Flu assay utilizes our proprietary real-time TMA chemistry to detect and differentiate SARS-CoV-2, influenza A and influenza B from nasopharyngeal and nasal swabs obtained ...
Result display time: 15 min
FLU A/B Antigen Test Kit (Colloidal Gold) is used for in vitro qualitative detection of influenza A/B antigen in human nasopharyngeal swab samples. PACK ...
... only a preliminary test result. The test is suitable for all people with common cold symptoms or for common cold screening. This test is suitable for Professional Use only. PACK FORMATS 1 ...
Result display time: 15 min
Sample volume: 0.1 ml
Specificity: 96.7 % - 99.7 %
COVID-19 & FLU A/B Antigen Test Kit (Colloidal Gold) is used for in vitro qualitative detection of 2019 Novel Coronavirus antigen and influenza A/B antigen ...
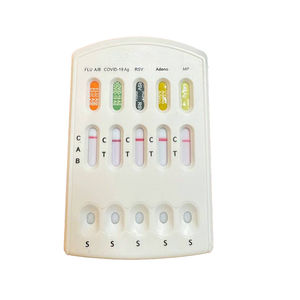
Result display time: 15 min
Multiple Respiratory Antigen Combo Test is only used for the intro qualitative detection of Multiple Respiratory Multipathogen Antigen SARS-CoV-2/Respiratory Syncytical Virus/Influenza AVirus/Influenza ...
SCREEN TEST COVID-19+FLU A/B Sars-Cov-2 Antigen + Flu A/B Rapid Test REF: SC-1262-20 The Covid-19 Antigen Rapid Swab and Influenza A + B ...
... diagnostic use. The RIDA®GENE Flu test, performed on the Roche LightCycler® 480II, is a multiplex real-time RT-PCR for the direct qualitative detection and differentiation of influenza virus RNA (influenza ...
Sample volume: 0.01 ml
... etc. - Package: 32 T/ kit, 96 T/ kit - Limit of detection: SARS-CoV-2: 200 copies/mL; Influenza A: 500 copies/mL; Influenza B: 500 copies/mL ...

Result display time: 15 min
Specificity: 99.9 %
Sensitivity: 99.9 %
Influenza is a highly contagious, acute, viral infection of the respiratory tract. The causative agents of the disease are immunologically diverse, single-strand RNA viruses known as influenza viruses. ...
Result display time: 120 min
NeoPlex™ FluCOVID Detection Kit simultaneously detects SARS-CoV-2, Influenza A and B virus in one-step real-time RT-PCR based on the TaqMan® technology. Feature of ...
Result display time: 270 min
Specificity: 100 %
Sensitivity: 97.6 % - 100 %
NeoPlex™ RV-Panel A Detection Kit simultaneously detects 10 viral respiratory pathogens in a single-tube multiplex real-time RT-PCR based on proprietary C-Tag™ technology. Feature of Product Single-Tube ...
Result display time: 10 min
The Influenza Antigen Quick is a qualitative immunoassay for the determination of influenza A and B virus antigen in human nasal and nasopharyngeal specimens. The Influenza ...

... eight (8) sample allotments in a 96-test kit, and robust stability are compatible with an existing workflow. Quidel wants to help any molecular laboratory use Lyra to expand their existing test ...
Quidel
... SARS-CoV-2 & Influenza A/B Detection Kit (Direct Fluorescence PCR) Sample Type: Oropharyngeal swab or Nasopharyngeal swab Assay Target : SARS-CoV-2, Flu A, Flu B ...
Zhejiang Orient Gene
... recommended the Respiratory Pathogens Multiplex PCR test for flu season. Positive results from this kits are indicative of the presence of SARS-CoV-2, Influenza A Virus (H1N1 / H3N2 / ...

Result display time: 90 min
Sample volume: 0.008, 0.02 ml
... remaining specific to the SARS-CoV-2 and Influenza types A and B genomes. NZYTech’s COVID-19 & Flu A/B Multiplex One-Step RT-qPCR Kit, IVD is a multiplex assay detecting ...
NZYTech

... single channel on real-time PCR instrument. Allplex™ Respiratory Panel Assays allows faster, more reliable and comprehensive test results than any other products by combination with Seegene’s automation platforms. KEY ...
Seegene

300+ items; RT-PCR; SARS-CoV-2; Variant; Omicron; Flu A, Flu B; RSV; Tuberculosis; Dengue; Respiratory Pathogens Multiple Detection Kit A variety of detection kits ...

lnfluenza A/B Fast Test Kit (lmmunofluorescence Assay) Intended Use lnfluenza A/B Fast Test Kit (lmmunofluorescence Assay) ...

... tend to peak (October through March in the northern hemisphere and April through September in the southern hemisphere), influenza is often the first virus people think of.3 In the US, the CDC estimates that 8% of the ...

Infectious diseases are extremely common worldwide, some of them strike more often than others. Many infectious diseases cause complications that can range from mild to severe, even some organisms can cause death too. YHLO can provide ...

... fluorescence immunochromatographic technology to detect influenza A and B virus antigens. Clinical application This kit is used for in vitro qualitative detection of influenza ...
Shenzhen Zijian Biotechnology Co., Ltd.
... Coronavirus, Influenza A and B Virus Nucleic Acid Test Kit This kit is intended for the qualitative detection of ORF1ab gene of 2019-Novel Coronavirus ...
Jiangsu Mole Bioscience
Your suggestions for improvement:
the best suppliers
Subscribe to our newsletter
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group


























Please specify:
Help us improve:
remaining