- Medical Technical Facilities >
- Healthcare IT, Telemedicine >
- Security software
Security software
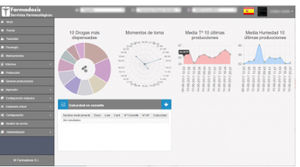
... ensuring a more comfort, security and efficiency control of the automation system in the pharmacy, being unique on the market, permitting the management of 15,000 patients in only 40 seconds, converting AMCO+ into the software ...
... Description
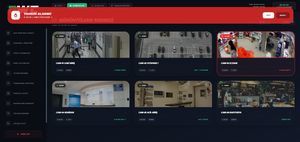
We consolidate traditionally separate
security layers under a single intelligent unified
software platform. The solution converts a monitored site into a proactively managed control center by enabling ...
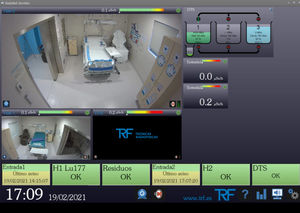
... RadyNet is the essential software for the comprehensive control of all detectors and devices manufactured by TRF. It allows remote management of radiation monitor networks GMRady and waste management systems DTS, also integrating video ...
Técnicas Radiofísicas S.L.U.

... Innovative software designed for specialists in the medical field who seek to work more efficiently, in an organized and cutting-edge way. Developed using the latest technologies and an intuitive interface, Medicrom simplifies daily ...
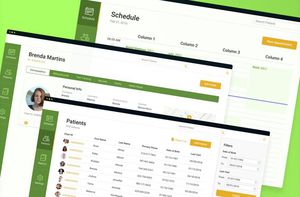
... high EHR usability, interoperability, compliance, and efficiency. Meet ScienceSoft, a Reliable EHR Software Development Company 35 years in software development and 19 years in healthcare IT. Working experience ...

... . Access and review consultation data, imaging, and records with an FDA-cleared DICOM viewer Data Security and Privacy Patient data security and privacy are the mainstaysof the United States Health Information ...
GlobalMed

... technical documentation of medical devices and for documentation processes for quality and information security management systems. With our software, you can digitize and link your processes, reduce effort and minimize ...


... needing to connect workstations to the internet or having to manually enter annoying login credentials. Additionally, the security of your institution remains intact, and your staff is relieved from extra work. The solution is called ...
Discus
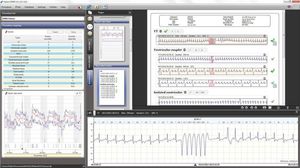
... conclusion templates make detailed and comprehensive reporting almost instant. Built on the Microsoft .Net Framework to ensure security, performance and compatibility are continuously optimised by Microsoft through their automatic ...
... subway or while you wait for your kids to finally get dressed. medikit is always there for you, everywhere! The cloud-based software not only works perfectly on a PC, but also on a smartphone and tablet. Everyone can download the medikit ...
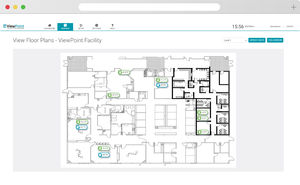
... controlled so you don’t have to worry about security, operating system, or software updates, and cloud system data is backed-up more regularly and stored longer without size restrictions. With cloud software, ...

... “BAS” Software is a software which is complaint with CFR 21 Data Integrity as data attends the data standards of ALCOA: Attributable as all individuals need to log in with a password Legible as data is easy to understand ...
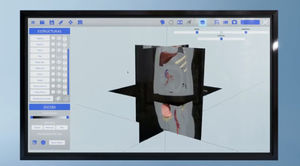
... Fluxus is the innovative clinical environment that represents a paradigm shift for oncology analysis in tumour progression and surgical planning. By assisting with cutting-edge technologies such as AI and D/L, Avamed Synergy enables healthcare professionals ...

... included in diagnostic and therapeutic decisions. The quality of care is thus increased and offers healthcare employees more security in their workplace. The individual components of the healthcare content management system ensure ...
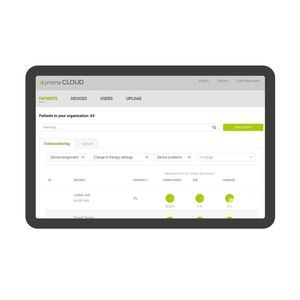
... monitored and adjusted remotely, not only with prisma sleep therapy devices, but also with prisma VENT. Data Protection and Security. Protecting your data is our top priority, especially when it comes to medical information. That's ...

... communications, alarm centralisation & system integration for estate management, site safety and smart cities Ensuring the security, safety and smooth operation of a large, complex premises is a challenge. There are often multiple ...
Multitone Elektronik International GmbH

... with showing the message sent to the said device while the device is online. Devices system update Support an Android security notification system patch update Device info display Including information of location of use, number ...
CILICO

... To meet your needs, an online StairSafe assistant is there to help. Included with StairSafe Safe and Secure Settings Maintain the safety and security of your details, as well as the information of your assigned ...
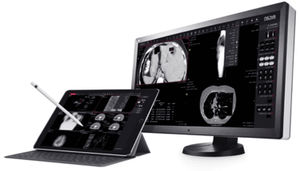
... streaming to the aycan viewer. Supports Lightweight Directory Access Protocol (LDAP), HTTPS Security integration as well as Single Sign-On (SSO) through security tokens to allow fast and flexible usage of the system ...

... information live streamed from our reader systems to your own personal cloud storage. BUILT WITH SECURITY IN MIND Our platform is built using the latest security tools and protocols to provide peace of mind to our ...
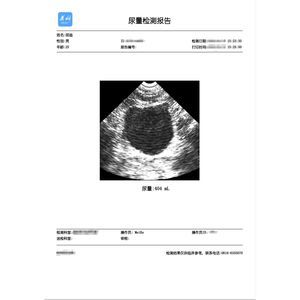
... Scope of application: This software is used for the storage, display and printing of bladder scanner detection results (grayscale image and urine volume data). Contraindications: None Features: 1. The software ...

... anti-collision, pinpointing the best fitting tags for the target application while offering security for proprietary branded tags and standards-based security for generic tags: designed for OEMs who build for high privacy ...

... A sophisticated Health technology software designed to optimise the Global Healthcare arena. Green Cube® facilitates the overall patient care, efficiency of any hospital, pharmacy, procurement centre, primary care, dental, ophthalmology, ...

... patients. Program menu The software enables creation and storage patient database . Additionally, we have the option of i importing and exporting research and creation research statistics for a given patient. To ensure security ...
the best suppliers
Subscribe to our newsletter
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group