- Surgery unit
- Surgical Instruments
- Arthroscopy electrode
- Baisheng Medical Equipment

- Products
- Catalogs
- News & Trends
- Exhibitions
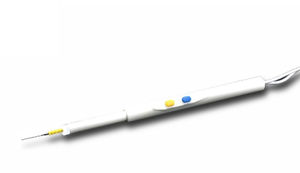
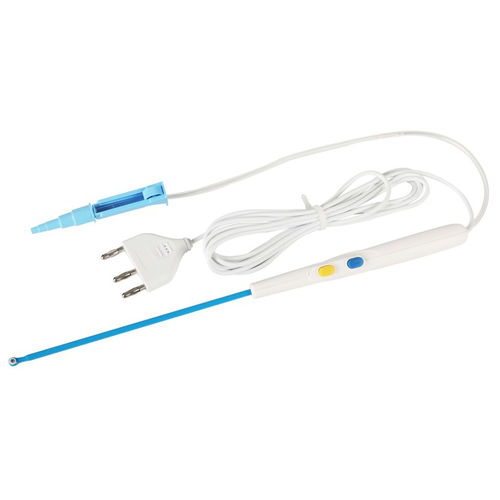
Electrosurgical electrode OBS-Dpcarthroscopicablationcoagulation

Add to favorites
Compare this product
Characteristics
- Applications
- electrosurgical, arthroscopic
- Procedure
- ablation, coagulation, tissue
- Type
- knife
- Zone of use
- subcutaneous
- Use
- disposable
Description
Match with Electrosurgical Generator(Unit), Radio Frequency Generator(Unit) and other High Frequency Equipment.
Type:
D: Disposable
These devices are single use supplied sterile. Check plug matching on generator base and connect. Press CUT/yellow button for cutting and COAG/blue button for coagulation. Maximum voltage is not be exceed 90KV peak
Product feature:
1.Intended for ablation,tissue modification and coagulation of soft tissue in arthroscopic procedures such as hip, shoulder, ankle, wrist, elbow and knee.
2.Extending shaft enables good visualization and easy insertion through subcutaneous tissue,and well insulated coating guarantee its safety and reliability.
3.Irrigation/suction solution is available with a cannula and an adjustable flow control valve.
4.Tips in different shapes and angles fit in particular arthroscopic procedure.
5.Compatible with most electrosurgical generators.
Warning
These devices should never be used when:
There is visible evidence of damage to the exterior of the device such as cracked or damaged plastic or connector damage.
These devices fail the inspection described herein.
In the presence of flammble gases, flammable prep solutions or drapes. Oxidizing. Gases such as Nitrous Oxide (N2O),or in oxygen-riched environments.
Do not used monopolar electrosurgery on small appendages, as in circumcision or finger surgery.
VIDEO
Exhibitions
Meet this supplier at the following exhibition(s):

Other Baisheng Medical Equipment products
ELECTROSURGICAL DEVICES AND ACCESSORIES
Related Searches
- Surgery forceps
- Grasping forceps
- Surgical electrode
- Stainless steel forceps
- Electrosurgical unit electrode
- Straight forceps
- Electrosurgical system
- Reusable forceps
- Reusable electrode
- Ablation electrosurgical unit
- RF electrode
- Coagulation electrosurgical system
- Surgical stapler
- Monopolar electrode
- Electrosurgical forceps
- Sealing forceps
- Disposable surgical stapler
- Straight electrode
- Tissue grasping forceps
- Disposable electrode
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.