- Company
- Products
- Catalogs
- News & Trends
- Exhibitions
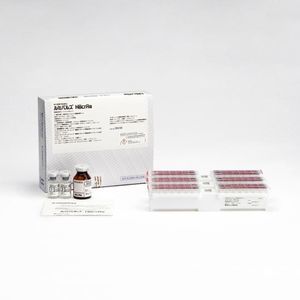
COVID-19 assay kit Lumipulse® Gfor antigensSARS-COV-2coronavirus

Add to favorites
Compare this product
Characteristics
- Applications
- COVID-19
- Tested parameter
- for antigens
- Micro-organism
- SARS-COV-2, coronavirus
- Sample type
- clinical, nasopharyngeal, saliva, laboratory
- Analysis mode
- chemiluminescence, enzyme immunoassay, CLEIA, molecular
Description
For in vitro diagnostic (IVD) use with the Lumipulse G system for detection and quantitative measurement of SARS-CoV-2 nucleocapsid protein antigen in human nasopharyngeal swab or saliva. As a diagnostic tool for the confirmation of a SARS-CoV-2 infection.
The assay utilises proven CLEIA (chemiluminescent enzyme immunoassay) technology with results that are available in up to 35 minutes. Please note that Lumipulse G assay need to be performed on either our LUMIPULSE G600II or LUMIPULSE G1200 in a clinical laboratory.
CE marked
Details
The 2019 novel coronavirus infection disease (COVID-19) is caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).1,2 In December 2019, the Health Commission of the City of Wuhan, Hubei Province, China, reported multiple pneumonia patients with unknown cause. On January 7th 2020, the World Health Organization (WHO) announced that the National Health Commission of China identified a new type of coronavirus, SARS-CoV-2.3 The WHO declared a COVID-19 pandemic on March 11th 2020 due to the worldwide spread of novel coronavirus infection.
To detect the virus, lower respiratory tract specimen, nasopharyngeal swab fluid and saliva of the patient are shown to be reliable samples for the detection of the SARS-CoV-2 virus.5,6 In general, the diagnosis of SARS-CoV-2 infection is made by molecular detection of the SARS-CoV-2 genes.
Assay principle:
Lumipulse G SARS-CoV-2 Ag is an assay system, including a set of immunoassay reagents, for the quantitative measurement of SARS-CoV-2 antigen in specimens based on CLEIA technology7 by a specific two-step immunoassay method on the LUMIPULSE G System.
VIDEO
Related Searches
- FUJIREBIO test kit
- FUJIREBIO solution reagent
- FUJIREBIO blood test kit
- FUJIREBIO serum test kit
- FUJIREBIO immunoassay test kit
- FUJIREBIO plasma test kit
- FUJIREBIO infectious disease test kit
- FUJIREBIO research reagent
- FUJIREBIO diagnostic reagent
- FUJIREBIO laboratory reagent
- FUJIREBIO protein reagent
- FUJIREBIO molecular biology test kit
- Enzyme reagent kit
- Whole blood detection kit
- FUJIREBIO respiratory disease test kit
- Histology reagent kit
- FUJIREBIO clinical test kit
- Optical assay kit
- Cassette assay kit
- Immunology reagent
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.