- Secondary care
- Oncology
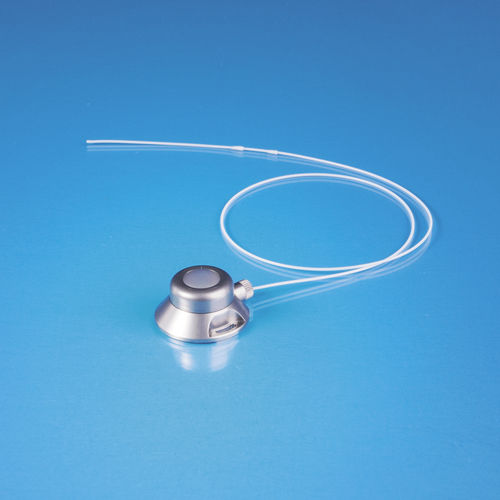
- Implantable venous port
- PakuMed Medical Products
Implantable venous port 111244 P-Narterialsingle-lumentitanium

Add to favorites
Compare this product
fo_shop_gate_exact_title
Characteristics
- Type of port
- venous, arterial
- Number of lumens
- single-lumen
- Material
- titanium
Description
TITAN-PORT V are totally implantable port catheter systems for venous implantation consisting of a titan chamber (port) with a self-sealing silicone membrane and a screw closure mechanism which enables the connection with the respective catheter.
Each system includes a 20 G access needle, a rinsing needle, a vein lifter, this instruction for use, a patient care guide and an implant card. If the port catheter system is a complete set (recognisable by the suffix “SET” at the end of the article number), it also includes a special port puncture needle SFN 0925 S, a tunnelizer and a matching introducer set (e.g., for implantation using the Seldinger technique).
The port catheter systems are placed on market as a procedure pack with difference in size of the port chamber, material and size of the catheter and associated introducer set.
The choice of size depends on the patient’s anatomy, the type of application, etc., and is at the discretion of the doctor.
TITAN port catheter systems A/V ensure repeated access to the central venous vascular system for the following applications, e.g.:
for long-term treatment of cytostatics agents and other “aggressive” medications
patients with poor peripheral veins requiring frequent intravenous injections
for infusion therapy
for parenteral nutrition
in HIV patients
for venous blood sampling
for blood transfusion
for high-pressure injections (systems marked accordingly are also suitable for high-pressure injection)
The advantage is a low risk of infection, simplified access and considerably improved quality of life for the patient as provided by a closed system.
VIDEO
Catalogs
No catalogs are available for this product.
See all of PakuMed Medical Products‘s catalogsExhibitions
Meet this supplier at the following exhibition(s):

Other PakuMed Medical Products products
TITANIUM PORT STANDARD
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.



