- Secondary care
- Cardiology
- Peripheral stent
- Bard Medical
Peripheral stent LIFESTENT® SOLO™
Add to favorites
Compare this product
fo_shop_gate_exact_title
Characteristics
- Type
- peripheral
Description
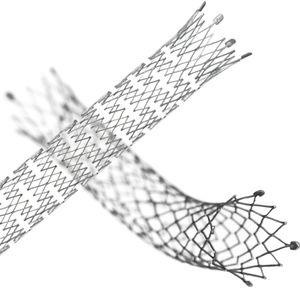
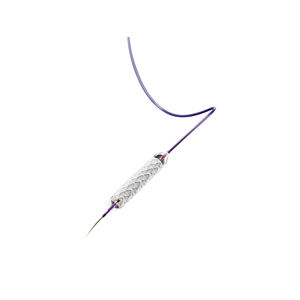
LifeStent™ Solo™ Vascular Stent System
FDA-approved for the SFA and full popliteal artery
Features:
Longest lesion length indication (up to 240 mm)1
Proven performance in long lesions2
Designed for enhanced distal and proximal stent placement accuracy
Treat longer lesions with one stent
About LifeStent™ Solo™ Vascular Stent
The LifeStent™ Solo™ Vascular Stent is the first commercially available 200 mm stent in the U.S. and potentially enables a single-stent solution. The LifeStent™ Solo™ Vascular Stent has sustained effectiveness up to three years in longer lesions. Furthermore, its enhanced delivery system is designed to reduce delivery system-dependent stent compression or elongation and enhance deployment accuracy. The LifeStent™ Vascular Stent Systems, in varying sizes, have been studied in more than ten clinical trials in the United States and globally.
The LifeStent™ Solo™ Vascular Stent is a peripheral stent intended to improve luminal diameter in the treatment of symptomatic de-novo or restenotic lesions up to 240mm in length in the native superficial femoral artery (SFA) and popliteal artery with reference vessel diameters ranging from 4.0 – 6.5mm. The LifeStent™ Vascular Stent is the only FDA-approved stent for the SFA and full popliteal artery. The LifeStent™ Solo™ Vascular Stent is available in 6 mm and 7 mm diameters and 200 mm in length.
Additionally, the LifeStent™ Vascular Stent is available in 5 mm, 6 mm, and 7 mm diameters; and 20 mm to 170 mm in length. To learn more about the LifeStent™ Vascular Stent,
Catalogs
No catalogs are available for this product.
See all of Bard Medical‘s catalogsRelated Searches
- Catheter
- Balloon catheter
- Stent
- Dilatation catheter
- Peripheral catheter
- Cardiac catheter
- Metal stent
- Catheter guidewire
- Diagnostic catheter
- Access catheter
- Venous catheter
- Central venous catheter
- Hydrophilic guidewire
- Blood vessel catheter
- PTA catheter
- Nitinol stent
- Triple-lumen catheter
- Self-expanding stent
- Vascular introducer
- Balloon catheter pump
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.