- Laboratory
- Laboratory medicine
- Flu test kit
- Medical Innovation Ventures

- Company
- Products
- Catalogs
- News & Trends
- Exhibitions
Infectious disease test kit GenoAmp® GRA2032for medical researchflupneumonia
Add to favorites
Compare this product
fo_shop_gate_exact_title
Characteristics
- Applications
- for infectious diseases, for medical research, flu, pneumonia, COVID-19, MERS
- Micro-organism
- coronavirus, influenza B, influenza A
- Sample type
- clinical, nasopharyngeal
- Analysis mode
- for PCR, for RT-PCR, for real-time PCR
Description
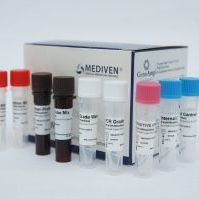
GenoAmp® Real-Time RT-PCR Flu A/Flu B/COVID-19/MERS-CoV
GenoAmp® Real-Time RT-PCR Flu A/Flu B/COVID-19/MERS-CoV
A real time molecular test for the screening and the differentication of Influenza A and B strains, COVID-19 and MERS-CoV
Product Code:GRA2032
Detection:
Flu A
Flu B
SARS-CoV-2
MERS-CoV
Specimen:
Nasopharyngeal swab
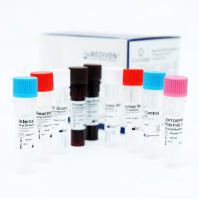
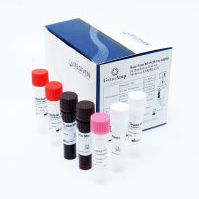
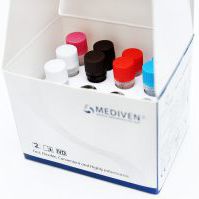
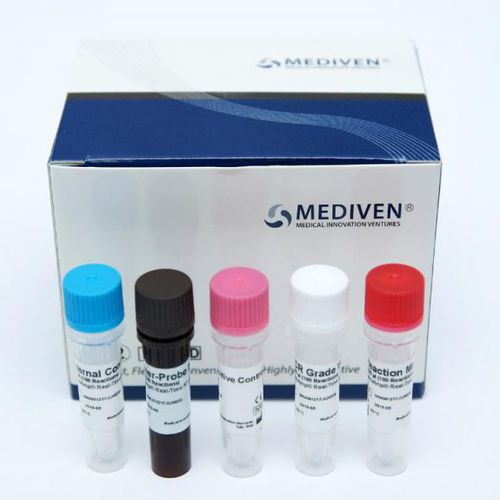
Kit Content:
Reaction Mix
Primer-Probe Mix
Internal Control
PCR Grade Water
Positive Control
Compatible Instrument:
Bio-Rad (CFX96)
LineGene 9600 Series
QuantStudio® 5 System
Qiagen Rotorgene-Q
Test per kit:100
Fast: 2.5 hours from sample to result.
Flexible: Includes internal control (IC) for monitoring of potential PCR inhibitions.
Convenient: Easy reaction setup and result analysis.
Highly Informative: Early detection of Influenza- Like Illenesses (ILI) disease cluster.
Catalogs
No catalogs are available for this product.
See all of Medical Innovation Ventures‘s catalogsOther Medical Innovation Ventures products
GenoAmp® Real-Time PCR Kits
Related Searches
- Assay kit
- Blood assay kit
- Serum assay kit
- Plasma assay kit
- Infectious disease detection kit
- Blood rapid diagnostic test
- Rapid lateral flow test
- Immunoassay rapid diagnostic test
- Molecular test kit
- Virus rapid diagnostic test
- Serum rapid diagnostic test
- Whole blood detection kit
- Respiratory infection test kit
- Plasma rapid diagnostic test
- Infectious disease rapid diagnostic test
- Clinical assay kit
- Whole blood rapid diagnostic test
- Lateral flow test kit
- COVID-19 detection kit
- Rapid respiratory infection test
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.