- Laboratory >
- Laboratory medicine >
- Respiratory disease test kit
Respiratory disease test kits
{{product.productLabel}} {{product.model}}
{{#if product.featureValues}}{{product.productPrice.formattedPrice}} {{#if product.productPrice.priceType === "PRICE_RANGE" }} - {{product.productPrice.formattedPriceMax}} {{/if}}
{{#each product.specData:i}}
{{name}}: {{value}}
{{#i!=(product.specData.length-1)}}
{{/end}}
{{/each}}
{{{product.idpText}}}
{{product.productLabel}} {{product.model}}
{{#if product.featureValues}}{{product.productPrice.formattedPrice}} {{#if product.productPrice.priceType === "PRICE_RANGE" }} - {{product.productPrice.formattedPriceMax}} {{/if}}
{{#each product.specData:i}}
{{name}}: {{value}}
{{#i!=(product.specData.length-1)}}
{{/end}}
{{/each}}
{{{product.idpText}}}

Result display time: 75 min
Real-time RT-PCR test for the qualitative detection of HA gene from Influenza A/B virus and N and ORF1a gene from SARS-CoV-2 in nasopharyngeal swab. Details Features: Hot Start PCR: high specificity Detection ...
Fujirebio

Result display time: 75 min
Real-time RT-PCR test for the qualitative detection of SARS-CoV-2 nucleic acids in various sample types. Details Features Hot Start PCR: high specificity Detection Target: N and ORF1a genes OneStep PCR: ...
Fujirebio
Result display time: 40 min
Real-time RT-PCR test for the fast qualitative detection of SARS-CoV-2 (N, ORF1a and RdRp) nucleic acids in various sample types. Details Features: Hot Start PCR: high specificity Detection Target: N, ORF1a ...
Fujirebio

... of multiplexing in this way are endless; allows complete patient profiling, reduces the amount of time spent on individual tests, identifies co-infections, differentiates between pathogens that exhibit similar symptoms ...

SARS-CoV-2 Array | 2-Plex Molecular Assay for SARS-CoV-2 and 1 other viral target Suitable for use with the Randox Discovery, the SARS-CoV-2 Array comprises 2 strains of coronavirus including SARSCoV-2 (COVID-19) and Sarbecovirus (confirmatory ...

... Hi-Plex and Lo-Plex testing. The VRI test can identify SARS-CoV-2 (COVID-19) and differentiate it from nine other respiratory infections with similar symptoms, including influenza and all known coronaviruses. Vivalytic ...

Result display time: 15 min
... self-collected by an individual aged 18 years or older or are collected by an adult from an individual younger than 18 years old. This test is intended for use in individuals with symptoms or other epidemiological reasons ...
Lepu Medical
... • ACCURATE • RELIABLE • EFFICIENT • CONVENIENT Principle: The test kit contains fluorescently-labeled 2019-nCoV Spike Protein (RBD), ACE2 Protein immobilized in test area ...
Lepu Medical

Result display time: 15 min
... qualitative detection. Colloidal Gold Antibody Test Kit Features • ACCURATE • RELIABLE • EFFICIENT • CONVENIENT Principle of Colloidal Gold Antibody Test Kit: This ...
Lepu Medical

... quickly deploy emergency tests for SARS-CoV-2 infection. Now, scientists can harness the flexibility and reproducibility of LC-MS to advance critical SARS-CoV-2 research. Waters SARS-CoV-2 LC-MS Kit ...
Result display time: 10 min
are both considered as highly contagious respiratory disease but triggered by different virus: SARS-CoV-2 and Flu A&B, respectively. GOLDSITE SARS-CoV-2 & Influenza A/B Antigen Kit (Colloidal ...
Goldsite Diagnostics Inc.
Result display time: 15 min
... considered as highly contagious respiratory disease. The COVID-19, Flu, RSV combined test is an option if you would like to determine what type of infection you have: ...
Goldsite Diagnostics Inc.

Result display time: 12 min
... -CoV-2 Antigen Kit (Saliva) is a lateral flow immunoassay intended for the qualitative detection of SARS-CoV-2 nucleocapsid antigen in saliva specimens from individuals who are suspected of COVID-19 by their healthcare ...
Goldsite Diagnostics Inc.

Result display time: 3 h
... testing. With Panther® Scalable Solutions, you can run over 1000 tests in 24 hours, attaining first results in about 3 hours. The Panther Fusion® SARS-CoV-2 assay is a real-time PCR test and the Aptima® ...

SARS-CoV-2 Nucleic Acid Detection Kit (Direct Real-time RT-PCR Method) Intended Use The Tianlong SARS-CoV-2 Nucleic Acid Detection Kit is intended for the qualitative ...
Xian Tianlong Science and Technology

Multiplex Detection Kit for SARS-CoV-2 Nucleic Acid and E484Q/P681R Mutants
Xian Tianlong Science and Technology

Product Name: SARS-CoV-2 Nucleic Acid Detection Kit (Real-time RT-PCR Method) Intended Use: Tianlong SARS-CoV-2 Nucleic Acid Detection Kit is intended for the ...
Xian Tianlong Science and Technology
Result display time: 15 min - 20 min
Sample volume: 0.01 ml - 0.02 ml
Specificity: 100 %
The Diagnostic Kit for SARS-CoV-2 1gM&lgG Antibody (Colloidal Gold) from KHB adopts the solid phase colloidal gold immunochromatographic technology for the qualitative determination of lgM&lgG antibody against SARS-CoV-2 ...
Result display time: 70 min
... Product Features Application: the test kit is an enzyme immunoassay for the in vitro qualitative determination of IgM or IgG antibody for SARS-CoV-2. Specimens: human serum and plasma ;High Throughput: ...
Result display time: 70 min
... Product Features Application: the test kit is an enzyme immunoassay for the in vitro qualitative determination of IgM or IgG antibody for SARS-CoV-2. Specimens: human serum and plasma ;High Throughput: ...

... The kit contains: -Freeze-dried master mix in amber glass vials (48 tests each) -Reconstitution Buffer tubes -Internal Control STAT-NAT® COVID-19 Positive Control kit is used ...

... This kit was developed and validated to be used with the following PCR instruments: Qiagen Rotor-Gene Q, BioRad CFX96 Touch™ Real-Time PCR Detection System, ABI 7500, ABI Quantstudio 5 and 7. Storage Conditions ...
... persons with signs and symptoms of respiratory infection. The RIDA®GENE Flu & SARS-CoV-2 test is intended to support the differential diagnosis of flu A/flu B and SARS-CoV-2 infections in patients ...
R-Biopharm AG

... SARS-CoV-2 test, which will be performed on the Roche LightCycler® 480II, is a multiplex real-time RT-PCR for the direct qualitative detection of coronavirus (SARS-CoV-2) RNA from human nasal/throat swabs of people with ...
R-Biopharm AG
... BAL). The RIDA®GENE Pneumocystis jirovecii multiplex real-time PCR is intended to use as an aid in diagnosis for respiratory infections caused by Pneumocystis jirovecii. General information: Pneumocystis ...
R-Biopharm AG
Result display time: 15 min
Sample volume: 0.1 ml
Specificity: 97.5 % - 99.6 %
COVID-19 Test Kit (Colloidal Gold Method) is a solid phase immunochromatographic assay for the rapid, qualitative detection of antigen to 2019 Novel Coronavirus in human nasal cavity. This test ...
Hangzhou Singclean Medical Products
Result display time: 15 min
Sample volume: 0.1 ml
Specificity: 96.7 % - 99.7 %
... -19 & FLU A/B Antigen Test Kit (Colloidal Gold) is used for in vitro qualitative detection of 2019 Novel Coronavirus antigen and influenza A/B antigen in human nasopharyngeal swab samples. PACK FORMATS 1 ...
Hangzhou Singclean Medical Products
Result display time: 15 min
FLU A/B Antigen Test Kit (Colloidal Gold) is used for in vitro qualitative detection of influenza A/B antigen in human nasopharyngeal swab samples. PACK FORMATS 1 Test/Box 10 ...
Hangzhou Singclean Medical Products
SCREEN TEST COVID-19+FLU A/B Sars-Cov-2 Antigen + Flu A/B Rapid Test REF: SC-1262-20 The Covid-19 Antigen Rapid Swab and Influenza A + B (nasopharyngeal swab) is a rapid immunochromatographic ...
Result display time: 10 min - 20 min
SCREEN TEST COVID-19 NT Sars-Cov-2 Neutralization Antibody Rapid Test REF: SC-1569-20 The SARS-CoV-2 Neutralizing Antibody Rapid Test Cassette (Whole Blood/Serum/Plasma) is ...

SCREEN TEST COVID-19 SALIVA Sars-Cov-2 Antigen Oral Fluid Rapid Test REF: 982735728 Disposable Self Test designed for the detection of the new coronavirus Sars-CoV-2 in human ...

Result display time: 10 min
... transmission of canine infectious diseases (canine distemper), and cause clinical symptoms such as conjunctivitis, pneumonia and gastroenteritis in dogs. High mortality, strong infectivity and short course of disease, ...

Result display time: 10 min
Sample volume: 0.0025 ml
... purpose】 Canine distemper virus (CDV) belongs to measles virus of para-mucosal virus family, which can cause severe infectious diseases in dogs. Detection of CDV IgG antibody in dogs can reflect the body's immune status. ...

Result display time: 10 min
Sample volume: 0.01 ml
... infectious rhinobronchitis, belonging to the herpesvirus A subfamily of the Department of Herpes Diseases. General clinical manifestations: The symptoms of upper respiratory tract infection are the main ...
Result display time: 5 min
... vitro quantitative determination of Krebs von den Lungen 6 in human serum, plasma. The test kit is used as an aid in the diagnosis of interstitial pneumonia disease activity. For in vitro ...

... accordingly, as an aid of diagnosis in clinic to test the IgG level in human sera can be very important. 【PRINCIPLE】 The kit integrates the method of immunological determination with immunochoromatograhy ...
HWTAi
Result display time: 15 min
Multiple Respiratory Antigen Combo Test is only used for the intro qualitative detection of Multiple Respiratory Multipathogen Antigen SARS-CoV-2/Respiratory Syncytical ...
HWTAi

Result display time: 5 min - 10 min
... operation, cloud platform quality control. 6. The detection sensitivity is high, comparable to that of enzyme-labeled kits. • Test devices • Droppers • Single Buffer • IFU
HWTAi

pneumonia detection kitcareGENE™ Pneumonia-12 Detection
INTENDED USE In vitro diagnostic medical device for diagnosing Pneumonia careGENE™ Pneumonia detection kit is a product that can qualitatively detect pneumonia-causing bacteria by amplifying pneumonia-causing ...
Result display time: 83 min
Sample volume: 0.01 ml
Specificity: 100 %
... diagnostic medical device for detection of COVID-19 careGENE™ COVID-19 RT PCR kit is an in vitro diagnostic medical device for qualitative detection of coronavirus disease (COVID-19) from RNA extracted ...
INTENDED USE In vitro diagnostic medical device for diagnosing Pneumonia careGENE™ Pneumonia detection kit is a product that can qualitatively detect pneumonia-causing bacteria by amplifying pneumonia-causing ...
Result display time: 75 min
Sample volume: 0.001 ml
Specificity: 100 %
... COVID-19 IgG is MicroDigital’s diagnostic kit for presumptive qualitative testing of antibodies to the SARS-CoV-2 virus, responsible for the COVID-19 pandemic, through the indirect ELISA method. With ...
MicroDigital Co., Ltd.

Sample volume: 0.01 ml
... Human GAPDH Anti-contamination: UNG-dUTP - Package: 32 tests/ kit; 96 tests/kit

Result display time: 15 min
Specificity: 99.9 %
Sensitivity: 99.9 %
... prevalent and are associated with most serious epidemies. [TEST KIT COMPOSITION] Individually packed test devices - Each device contains a strip with colored conjugates and reactive ...
Safecare
Result display time: 120 min
NeoPlex™ FluCOVID Detection Kit simultaneously detects SARS-CoV-2, Influenza A and B virus in one-step real-time RT-PCR based on the TaqMan® technology. Feature of Product Single-Tube Multiplex ...
Genematrix Inc.
Result display time: 45 min
Sample volume: 0.005 ml
Single-Tube Real-time RT-PCR Assay Ease of Use Automated analysis with NeoPlex™ Viewer software One-Step RT-PCR without separate cDNA synthesis step Whole process monitoring with internal control UDG System to prevent carry-over contamination
Genematrix Inc.
Result display time: 120 min
NeoPlex™ COVID-19 Detection Kit simultaneously detects SARS-CoV-2 in one-step real-time RT-PCR based on the TaqMan® technology. Feature of Product Single-Tube Multiplex Real-Time RT-PCR Assay Ease ...
Genematrix Inc.
Sample volume: 0.05, 0.025 ml
... Clinicians often detect multiple inflammatory markers at the same time, which is of great help to improve the accuracy of disease diagnosis. Clinical Significance Quantitative determination of hs-CRP (high-sensitivity ...
Autobio Diagnostics

This comprehensive genome-wide bovine genotyping array kit features over 777,000 SNPs, and is compatible with any breed of beef or dairy cattle. Overview The BovineHD BeadChip is a comprehensive genome-wide bovine ...
Illumina, Inc.

Result display time: 50 min
Sample volume: 0.01 ml
CE-marked Enzyme Immunoassay (ELISA) kit for the detection of IgM antibodies to SARS-CoV-2 in human serum. Main Features: - Number of tests: 96 - Principle: Indirect ELISA using ...
ERBA Diagnostics Mannheim
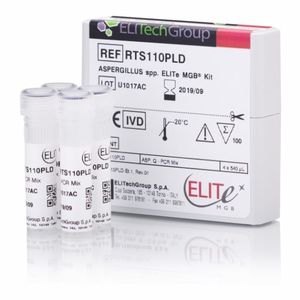
Result display time: 2 min
Specificity: 98 %
Sensitivity: 100 %
... lung diseases are at higher risk of developing severe outcomes. Early detection and quantification of Aspergillus with molecular methods is crucial to isolate infected patients and provide an appropriate treatment. Aspergillus ...
ELITech Group
Your suggestions for improvement:
the best suppliers
Subscribe to our newsletter
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group

























Please specify:
Help us improve:
remaining