- Laboratory >
- Laboratory medicine >
- PCR test kit
PCR test kits

... This kit is intended for the analysis the qualitative detection of Monkeypox Virus nucleic acids from a specimen(such as skin lesions) through Real-Time Polymerase chain reaction ( PCR). Features Enhanced Sensitivity ...

... been integrated into the kit in order to check PCR inhibition and extraction. The amplification data of the internal control is detected with the HEX filter. The internal control can be added either during RNA extraction ...
Anatolia Tani ve Biyoteknoloji Urunleri Sanayi ve Ticaret A.S.

Result display time: 60 min
... Complexity Similar workflow to AmplideX PCR/CE FMR1*† kit eases implementation and training Multiplexed, scalable design allows analysis of single-nucleotide variants, small indels, and copy-number changes from a single ...
Asuragen
Result display time: 30 min - 40 min
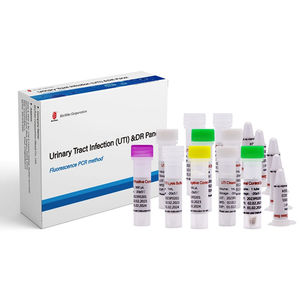
... Urinary tract infection is a common high incidence disease worldwide, it has become the second largest infectious disease after respiratory tract infection. It’s high morbidity and mortality is a serious threat to human health and life safety. Diagnosis ...
Bioteke Corporation

Sample volume: 2 ml
... 50 tests/ kit 【Expected Usage】 This kit is suitable for the detection of avian influenza virus (AIV) RNA in throat swab,milk and whole blood samples, and the test results can be ...

... and laboratory finding. RTA HBV Real-Time PCR Kit has been validated for use with RTA Viral DNA Isolation Kit or RTA Viral Nucleic Acid Isolation Kit for its best performance. RTA ...

... AZFa, AZFb and AZFc Part no. 800.001-32 Ready to use Add only DNA. Ready for PCR. Taq polymerase included No. of tests / Kit size 32 tests for AZFa, b, c – basic set 10 tests ...

Non-small cell lung cancer (NSCLC) patients with EGFR mutations can achieve good therapeutic effects by using epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs) such as gefitinib, erlotinib, and osimertinib. However, primary/acquired ...

Result display time: 120 min
... quick and exact diagnosis of infected people combined with quarantine measures. The AID SARS-CoV-2 Detection Kit is a Real time PCR screening kit for the fast and sensitive detection ...
Result display time: 35 min
... for canine health care. Recently, real-time PCR is being used for canine skin disease detection, enabling the simultaneous detection of all significant viral targets. CareDx™ Canine Skin Disease Real-time PCR Kit ...
Genesystem Co., Ltd.
... The SARS-CoV-2 Real-Time RT- PCR Assay is designed for specific and qualitative detection of SARS-CoV-2 RNA in samples such as oropharyngeal swabs, nasopharyngeal swabs or sputum suspected of SARS-CoV-2. ...

... AICON-BIOTM Feline Coronavirus (FCoV) Real-time PCR Kit utilizes real-time PCR technology and premixed freeze drying method to accurately and specifically detect Feline Coronavirus (FCoV) nucleic acid ...

... 1.A multiplexed test 2.Accuracy and reliability of the results 3.Low sample input Features 1. Simultaneous detection of seven monomorphic homopolymer biomarkersassessment 2. Fast results – 1 hourfrom sample to result 3. ...

... Specimen: Nasopharyngeal swab Kit Content: Reaction Mix Primer-Probe Mix Internal Control PCR Grade Water Positive Control Compatible Instrument: Bio-Rad (CFX96) LineGene 9600 Series QuantStudio® 5 System Qiagen Rotorgene-Q Test ...
Medical Innovation Ventures

... Hepatitis B virus (HBV) nucleic acid detection kit (fluorescence PCR method) Features: - For in vitro quantitative detection of HBV DNA in human serum or plasma ...
Guangzhou Biotron Technology Co.,Ltd
... 2019-nCoV/IAV/IBV Nucleic Acid Test Kit ( PCR- fluorescence probe method) This kit is a real-time RT- PCR test intended for the qualitative detection ...
hecin-scientific

... of cancer to facilitate targeted therapy. By incorporating Droplet Digital PCR technology, Droplex shows improved sensitivity with wider mutation coverage. Droplex Test Series will provide the possible course of ...
Gencurix
... amplification of selected DNA fragment with the detection of PCR products in real time. The PCR kits of the “MOLgen” series include a Positive Control sample (PC) corresponding to a given PCR ...

Mutations in the epidermal growth factor receptor (EGFR) gene are predictive of the response to EGFR tyrosine kinase inhibitor (TKIs) therapies. The ViennaLab StripAssay® identifies 30 EGFR mutations relevant for non-small cell lung cancer (NSCLC) TKI ...

... Intended use: Detection of SARS-CoV-2 specifie RNA Test principle: Real-time reverse transcription polymerase chain reaction and fluorescent probe-primer Sample: Nasopharyngeal swabs(NPS) and sputum Store temperature: -25 °C to -15 ...

Sample volume: 0.1, 0.4, 1.5, 1 ml
... cervical caner. The test results are only for clinical reference, not for case confirmation or exclusion. Test Principle: The specific primers and specific fluorescent probes are designed for the conserved region ...
Biokey Health
Result display time: 10 min - 15 min
Specificity: 98.8 %
Sensitivity: 100 %
... most accurate and reliable tests in the world There is a critical, global need for serology assays that can complement nucleic acid ( PCR) tests for diagnosing COVID-19 infection. Although critically ...

... principle and method This kit designs specific primers and probes for the highly conserved regions of monkeypox virus. In the presence of genome templates of Monkeypox Virus in the reaction system, PCR reaction is performed ...
Shenzhen Zijian Biotechnology Co., Ltd.
... Nucleic Acid Detection Kit ( PCR Fluorescence Probe Method) is used for the in vitro qualitative detection of 2019 novel coronavirus (2019-nCoV) RNA in upper respiratory tract specimens (nasopharyngeal ...
... with TKI resistance, mutations T790M exon 20 result in TKI resistance in about 50% of patients. [intended use] iThis kit is used for qualitative detection of paraffin-embedded pathological tissue or sections, fresh tumor tissue ...
Jiangsu MicroDiag Biomedicine Technology Co., Ltd.
the best suppliers
Subscribe to our newsletter
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group