- Company
- Products
- Catalogs
- News & Trends
- Exhibitions
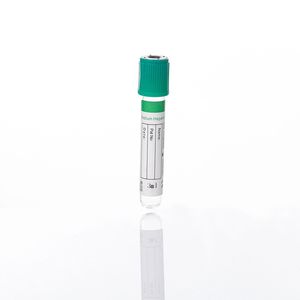
Microbiology test tube for blood glucose analysisfor hematological analysiswith flat-bottom

Add to favorites
Compare this product
fo_shop_gate_exact_title
Characteristics
- Applications
- microbiology, for blood glucose analysis, for hematological analysis
- Shape
- with flat-bottom
- Sample type
- capillary blood
- Material
- glass
Description
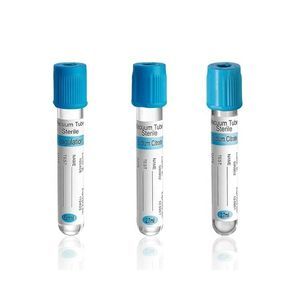
Medical Non-Heparinized Micro-hematocrit 75mm Hematocrit Blood Capillary Tube
Micro-haematocrit capillary tubes are made of glass and are available in different lengths and diameters. The standard length is 75 mm. The standard inside diameter is 1.1-1.2 mm and the standard outside diameter is 1.5-1.6 mm. 100 capillaries are packed in vials, 500 vials are in a carton.Supplied with color-coding on the packaging and on the tubes in plain (blue color), or with uniform sodium heparinisation (red color).
Medical Non-Heparinized Micro-hematocrit 75mm Hematocrit Blood Capillary Tube
Micro-haematocrit capillary tubes are made of glass and are available in different lengths and diameters. The standard length is 75 mm. The standard inside diameter is 1.1-1.2 mm and the standard outside diameter is 1.5-1.6 mm. 100 capillaries are packed in vials, 500 vials are in a carton.Supplied with color-coding on the packaging and on the tubes in plain (blue color), or with uniform sodium heparinisation (red color).
VIDEO
Catalogs
No catalogs are available for this product.
See all of HWTAi‘s catalogsRelated Searches
- Sample tube
- Container
- Laboratory sample tube
- Microtiter plate
- Collection tube
- 96-well microplate
- Sample box
- Pipette tip
- Cylindrical collection tube
- Laboratory holder
- Laboratory microplate
- Pipette
- Blood collection tube
- Laboratory vial
- Polypropylene sample tube
- Tube laboratory rack
- Waste container
- Sampling kit
- Conical test tube
- Collection tube with anticoagulant
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.