- Laboratory >
- Laboratory medicine >
- Respiratory disease test kit
Respiratory disease test kits

Result display time: 75 min - 80 min
Sensitivity: 96.4 % - 99.5 %
... Information Registration certificate: CE:Ref. No.:GZ 8821-2020; China NMPA: GUOXIEZHUZHUN 20203400229 Specification: 48 tests/ kit;96 tests/ kit Amplification time: 1h20min Instrument ...
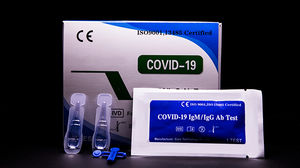
Result display time: 10 min - 15 min
Sample volume: 0.01 ml
Specificity: 99, 100 %
... COVID-19 IgM/IgG Ab Test is used for qualitative detection of the IgM and IgG antibodies of COVID-19 in human serum/plasma or whole blood. ...
Beijing Aeonmed

Result display time: 10 min
Specificity: 99 %
Sensitivity: 91 %
... approved by the competent authorities. The proven test results are excellent: The clinical sensivity is 91 %, the clinical specifity is 99 %. PCL Saliva Test Single 3 in 1 The test consists of: - ...
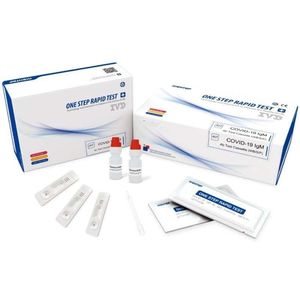
... TEST PRINCIPLE This kit is based on the principle of gold label immunochromatographic test and uses capture method to detect the COVID-19 IgM antibody in the sample. When the sample contains the COVID-19 ...

Result display time: 15 min
... The Influenza A+B Test detects and differentiates influenza type A and type B antigens directly from nasal swab and nasopharyngeal swab specimens. A single sample can be used to run both the Influenza A+B Test and RSV ...
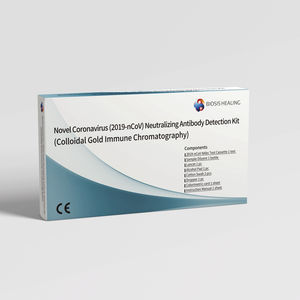
Specificity: 97.9 %
Sensitivity: 96.7 %
... Neutralizing Antibody Detection Kit (Colloidal Gold Immune Chromatography) To evaluate the level of neutralizing antibodies in humans after being vaccinated with 2019-nCoV vaccine and recovering from infection. The ...
Result display time: 15 min
... trusted, accredited, accurate & easy to use. • Guaranteed results in 15 minutes. • Fully Certified: TGA Supervised & Self- test approved; fully CE Certified. ...

... Extraction Vial five times just before applying the sample to the Test Strip. 7.Dispense 80µL (3 drops) of the specimen into the sample well on the cassette, and start the timer. 8.Interpret the test results at 10~15 ...

... Multiplex qPCR Detection Kit is an in vitro diagnostic test, based on real-time polymerase chain reaction technology. It tests for the presence or absence of ribonucleic acid (RNA) of ...
Result display time: 10 min
... 1 × 5 Bags, each bag contains 1 test cassette and 1 desiccant 1 × 5 Extraction tube 1 × 5 Extraction tube nozzle 1 × 5 Sample Extraction Tube Bottle 1 × 5 Individually packaged sample for sampling the sterile swab 1 Printed operating ...
... Use for COVID-19 test : Stable preservation of virus from nasopharyngeal swab sample This Sample protective liquid can rapidly inactivate virus particles in a sample to maximise the safety of frontline medical workers. The sample preservative ...
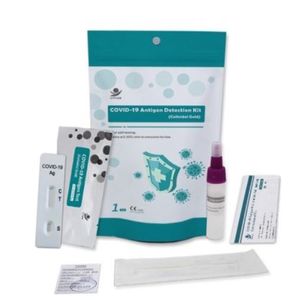
... COVID-19 Antigens Test Kit (Colloidal Gold) - Coronavirus ...
the best suppliers
Subscribe to our newsletter
Receive monthly updates on this section.
Please refer to our Privacy Policy for details on how MedicalExpo processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group














